Electro Carcinoma Treatment Device Electro Carcinoma Treatment can be done with external pad electrodes if the tumor is close to the skin (within 1+1/2") via the DC Electrifier with 5mA current for up to 4 hours daily for 2-3 weeks, or by a medical professional with the ECT Device with needles in tumors using up to 50mA for 30 minutes or more which is often enough to kill the cancer cells in the area between the electrodes with one treatment. With this ECT Device needle electrodes are placed through the skin into the center or at the edge of the targeted cancerous tissue. A selected electrical current is then sent to the electrode array, causing permanent damage to the cancerous cells. The dead cells are left in the body to be removed by the body's natural immune system. ECT potentially offers significant advantages over radiofrequency and cryoablation, the two leading thermal ablation technologies in the market today. The advantages include:
Clearly defined and predictable treatment margins.
Complete destruction of tissue adjacent to large blood vessels (no heat sink effect). This is the same treatment modality that is used throughout all of China now as an inexpensive and effective way to kill tumors. Researchers found that the current can stop or kill tumors by these means; changing the PH in tissues close to the electrodes to kill the tumor, changing an enzyme that the cancer cells need to reproduce, toxifying the tumor with oxygen that is produced by electrolysis, changing the transmembrane voltage in the cancerous cells, producing tumor-damaging toxins from the electrochemical reactions, and stimulating the cellular and humoral components of the immune system. Electrodes: You will be supplied six 3” long electrodes which are PTFE insulated (which makes it hydrophobic and slick) and have a 12 degree tip. This insulation needs to be scraped off with a razor blade to expose enough needle metal to equal the expected depth of the tumor where the needle will be inserted. A topical pain killer is helpful to lessen the pain of puncturing the skin. Click here for ECT Device usage instructions. 
Additional Therapy:
Most cancer patients are acidic and hypoxic (low oxygen), which is evidenced by a higher-than-normal
breathing rate. So first the patient needs to alkalinize their body with grape juice fasting to start, then a near-vegetarian diet with alkalinizing
supplements. This acts to stop cancer growth and spreading. The hardest part of fighting cancer is sticking to the alkalinizing diet. The patient will need to stick to it as much as is humanely possible. This will retrain his/her eating habits to make sure there is not a recurrence of cancer later.

SCIENTIFIC STUDIES: from http://www.iabc.readywebsites.com/page/page/623960.htm:
Five year survival rates for liver cancer patients treated in China is approximately 15%, whereas the five year survival rate for liver cancer patients treated with conventional therapies in the U.S. is approximately 5%. Dr. Nordenström's early five year survival rates for advanced stage breast cancer patients was approximately 60%.
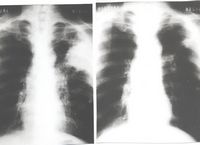
The X-ray images (from: Journal of the International Association for Biologically Closed Electric Circuits in Medicine and Biology, Vol. 1, January-December, 2002), for a 52 year old lung cancer patient show a 9.5 cm by 11 cm carcinoma (left photo), diagnosed by needle biopsy. Six platinum electrodes were inserted into the skin and into the tumor mass using X-ray monitoring. After the patient received six months of electrical treatment (EChT), the tumor completely disappeared (right photo). The patients progress has been very good.
In 1987, Dr. Björn Nordenström introduced BCEC and EChT to the Chinese medical profession. Since that time, considerable progress has been made. Dr. Xin Yu-Ling, Head of Thoracic Surgery at Friendship Hospital in Beijing, China and his staff have administered many EChT treatments. The Cancer Center of P.L.A., Nanjing Ba-Yi Hospital, Nanjing, China also treats cancer patients using EChT. EChT is also available at Guangxi Cancer Institute and Hospital, Guangxi, China.
The Journal of the IABC (Vol. 1, January-December, 2002) provides an overview of the results and therapeutic efficacy for EChT, alone, or in combination with other cancer therapies. In his paper, "Clinical Effectiveness Report for Approximately 11,000 cancer Patients With Various Kinds of Tumors Treated With Electrochemical Therapy" (EChT), Dr. Xin, Yu Ling has reported some impressive results. Most of the patients treated had one of the following forms of cancer: esophageal cancer, lung cancer, liver cancer, skin cancer, breast cancer, cancer of the head and face and metastatic lymph node cancer. Almost 70% of the tumors treated were larger than 5 cm.
The five year survival rate for EChT treated cancer patients has been approximately 69% for the combined stage I and stage II categories. If the large numbers of stage III Chinese cancer patients, with very large diameter tumors are included, the five year survival rate is 53%.
Many European cancer patients have been treated with EChT in various European hospitals and clinics including the Klinik St. Georg, Bad Aibling, Germany and Karolinska Hospital, Stockholm, Sweden.
In an Bioelectrochemistry article " Treatment of Tumors" details of the Chinese study results were listed as such: 5 year survival rates after having received ECT | Malignant tumors treated | % survival rate | | skin cancer | 80% | | laryngeal cancer | 62% | | tongue or lip cancer | 62% | | prostate cancer | 50% | | lung cancer | 39% | | jaw + facial tumors | 39% | | Benign tumors treated | % survival rate | | thyroid tumors | 99% | | prostate tumors | 71% | | thyroid carcinoma | 53% | | breast cancer | 50% | | thyroid carcinoma | 53% | | chest-abdominal wall metastases | 43% | (These percentages are not in conflict with the 30% rate of complete tumor elimination that the article by the Marburg Institute lists because they aren't percentages of complete tumor elimination, but rather of percentages of patients still alive after 5 years.)
Ablation
of Neoplasia by Direct Current
Baylor college of Medicine, Houston TX
British Journal of Cancer
1994, vol. 70, no2, pp. 342-345 (14 ref.)
The application of low-voltage direct electrical current (DEC) has been studied in an humans for the ablation of anal condylomata, oesophageal cancer and Kaposi's sarcoma. Twenty milliamps of DEC passed through multiple 6 cm×1 cm, flat-plate longitudinal electrodes into the squamous mucosa of the oesophagus of healthy dogs for periods ranging from 10 min to 2 hr resulted in denudation and necrosis [death] of the oesophageal mucosa at the site of application of the current. In humans, the application of DEC to two patients with benign anal condyloma acuminara, three patients with inoperable obstructing oesoghageal cancer and one patient with disseminated Kaposi sarcoma resulted in striking necrosis of tumour tissue that was confirmed by macroscogic and microscopic studies.
ECT summary from patent
6738663 Tumor cells are more sensitive to changes in their microenvironment than are normal cells. The effect of the application of direct current to cells with platinum electrodes has been summarized succinctly by Li et al.:
Water migrates from the anode to the cathode while fat moves in the opposite direction (this migration causes local hydration around the cathode and dehydration around the anode).
The tissue becomes strongly acidic at the anode and strongly alkaline at the cathode.
The distributions of macro- and microelements in the tumor tissue are changed.
Protein is denatured in the electrochemical process (hemoglobin is transformed into acid hemming around the anode and alkaline hemming around the cathode).
Chlorine, which is a strong oxidant, is liberated at the anode, whereas hydrogen, which produced local cavitation in the tissue, is liberated at the cathode.
By means of DC delivering adequate electric charge, a series of biological and electrochemical reactions take place in tissue. The cell metabolism and its existing environment are severely disturbed. Both normal and tumor cells are destroyed rapidly and completely in this altered environment.
Berendson et al. believe that the toxic properties of the chlorine close to the anode and of the hydrogen chloride within a broader zone may be enough to explain the clinical effects of ECT and that the liberated hydrogen ions determine the extension of the locally destroyed zone around the anode. Several researchers have also observed that destruction occurs around both anode and cathode (Song et al., Matsushima et al., and Xin et al.) as well as within the electric field established between them. (In early works Nordenstrom cautions against making the center of the tumor the cathode as it will cause concentration of the acidity at the wrong location but later reports that, in some cases, better results were achieved with the cathode at the tumor.) Subsequent work in Asia found an advantage in locating both electrodes within the tumor (Xin, 1997). Nordenstrom believed that the electro-osmotic transport of water compresses capillaries and was seen to block large pulmonary arteries in dog experiments. He points out that a sufficiently long interval of vascular obstruction will seriously interfere with the living conditions of the tissues. Thus, primary tumor destruction is obtained, along with a change in surrounding conditions that prevent the tumor from living. ECT is also believed to enhance the immune system of the patient (Chen et al., Chou et al). In studies conducted in mice there was infiltration of lymphocytes in tumor tissue six days after treatment. Leukocytes have a negative surface charge and are known to be sensitive to low voltage changes and changes in pH and ion strength. At an electrode voltage as low as 100 mV leukocytes concentrated at the anode. Many leukocytes can be attracted to the anode at relatively low voltages but are massively destroyed in the anodic field at 10 V. Nordenstrom recognized that electrophoretic movements will take place at low voltages and current densities and he discussed possible tissue changes with, for example, 10V and 1 to 2 microamperes applied for 30 days. He wrote ". . . it seems likely that DC treatment should be most beneficial when the technique approaches the mechanisms of closed circuit transport in spontaneous healing. This consideration implies the use of energies perhaps in the range of a few volts and a few microamperes over long time periods." He also deduced that AC potential may be used to heal tissue.
Procedurally, Nordenstrom used electrodes such as those shown in FIG. 1. The electrode is introduced through the chest wall (in the case of lung tumors) into the patient under guidance of biplane fluoroscopy or computed tomography under local anesthesia. In FIG. 1a hooked electrode ends 1 of platinum strings protruding from plastic tube 2 expand within tumor 3 to retain the electrode inside the tumor. In FIG. 1b platinum tubes 10-12 provide a larger surface area and can be chosen to correspond with the size of the tumor. Screw 14 is used to obtain biopsy tissue samples. The electrode 13 is shown implanted in tumor 20 in FIG. 1c. Tube 21 is constructed of Teflon.RTM.. Alternatively, FIG. 1d shows a tapered platinum tube 30. Screw 31 is used to obtain tissue for biopsy. Area 32 consists of collapsed wings which, as shown in FIG. 1e, expand 40 to stabilize electrode 30 in the tumor. Nordenstrom recognized that a platinum electrode can be improved mechanically by adding iridium. He stated some guidelines for electrode design and implantation. The electrodes should present a large surface area but must be easily introducible without causing too much damage. He recognized in 1994 that regression of cancer can take place both around the anode and the cathode in the tumor. Placement of both electrodes within the tumor can lead to a treatment result comparable with an initially successful surgical removal of a cancer. However, as with surgical removal, metastases may later start growing in the tissue around the former tumor site. Positioning the anode and cathode far enough away from each other will create a distant field effect that should prevent future metastases. Thus, he believed that ECT of "small resectable" cancers might be more efficient than conventional surgical resection. He advised that the use of multiple anodes and cathodes might cause an uneven distribution of current and recommended that electrodes be neither very close nor very far away from one another. The anode should be kept away from direct contact with large blood vessels if using the large currents and voltages used by Nordenstrom (but not with microampere level currents). The cathode may be placed in a blood vessel. Nordenstrom used a catheter that could be percutaneously inserted by Seldinger technique in, for example, a pulmonary artery. Electrodes can theoretically be placed on the skin (although he cautions against this in a later paper) or inserted through a chest wall, via a systemic artery, a systemic vein, a bronchus or in the pleural space. The venous routes and pleural space provide pathways for current that include the lymphatics. Nordenstrom also noted that flushing the anodal electrode with a charged agent such as Adriamycin or 5-fluoracil in a manner that causes even distribution of the drug with high concentration can lead to a remarkable regression and palliative effects of even large, incurable cancers. Whether supplied intravenously or orally, these two agents are attracted to the electrode, when given opposite polarity.
Nordenstrom reported treatment of 26 inoperable cancers of the lung in 20 patients starting in 1978 and followed up for 2 to 5 years. Twelve of the cancers were arrested and no fatalities occurred. He observed that in some cases multiple other small metastases in the lung parenchyma, distant from the sites of the electrodes, also appeared to regress after treatment of the larger metastases. He pointed out that the therapy was unoptimized at that time. Radiation treatment of lung tumors is not very effective. A rapid decrease in size of a poorly differentiated tumor after radiation treatment is often accompanied by re-growth of the tumor after a short time. Then the tumor is often more insensitive than previously to any attempts at a repeat course of radiation treatment. He foresaw an advantage of DC current treatment of primary neoplasms in the most surgically inaccessible locations such as the brain, spine, pancreas, liver and prostate and in patients who have been rejected for surgery, radiotherapy or chemotherapy because of poor general condition, cardiorespiratory insufficiency, diabetes mellitus, multiple locations of pulmonary metastases or failing response to chemotherapy. In a later report he cited favorable results with breast and bladder cancer. Also, he treated 14 patients with otherwise incurable cancers with ECT and a chemotherapeutic agent Adriamycin infused into the tumor. The principle, already mentioned above, is that an intramuscularly electropositive compound will be electrophoretically attracted to a neoplasm electrode given opposite polarity. This treatment was successful on larger tumors than was ECT alone and, in one case, abolished chronic cancer pain. Electrophoresis caused even distribution of the Adriamycin throughout the tumor, an effect probably not obtainable with injection.
Recent Human Results in Asia
B. E. Nordenstrom introduced electrochemical therapy in China in 1987 and, partly because of its relationship to traditional Chinese medicine (e.g., acupuncture), its use has been growing in China and interest has spread to Japan and Germany. Xin reported that, by 1994, 4081 malignant tumor cases were treated using ECT in 818 Chinese hospitals including esophageal, breast, skin, thyroid and liver cancers, as well as leg sarcomas. By the end of 1994 more than 6000 cases had been treated. Benign tumors such as heloid, angioma and freckle have also been treated.
Xin et al. published the results of treatment of 386 patients with lung cancer between 1987 and 1989. They found that damage of normal tissue could be eliminated by placing both electrodes into the tumor with anodes in the center and cathodes on the periphery. This has also enhanced the therapeutic effect significantly. They also concluded that the effect of ECT with lower current and longer treatment time is better than high current and shorter time.
Matsushima et al. and Chou et al also placed both electrodes inside the tumor. Matsushima et al studied 26 patients with 27 malignant tumors. The main complications were pain and fever for a few days after treatment. Pain during treatment, especially when the lesion was located in the neck or in soft tissue under the skin, was probably due to sensory nerve stimulation by the direct current. Some lung cancer patients had haemoptysis and pneumothorax.
Song et al. reported the treatment of tumors on the body surface with good results. ECT was found to be suitable for patients at great operative risk, for those who refuse surgery, for those who have not been cured by other means, and for those who have tumor recurrence. They discovered that metastatic enlarged lymph nodes can dissolve when the primary tumor is destroyed by ECT. The method was found to be simple, safe, effective, and readily accepted by patients. ECT can be used in primary as well as metastatic tumors, although the effect is better for primary tumors.
Lao et al. reported on the treatment of 50 cases of liver cancer using ECT. The indications for treatment were: the neoplasm was too large to be easily resected; it was unresectable because of location at the first or second hepatic portals; poor liver function secondary to severe cirrhosis making the patient unfit to stand the trauma caused by surgery; cancer infiltration of visceral organs such as the diaphragmatic muscle, peritoneum, or lymph nodes at the hepatic portals.
Quan discussed the ECT treatment of 144 cases of soft tissue and superficial malignant tumors. Short-term effectiveness of treatment was 94.5% for tumors with a diameter of less than 7 cm. and 29.4% for tumors with a diameter of more than 7 cm. He found that the earlier the stage the more effective the treatment and that ECT for malignant melanoma is more effective than chemotherapy and no different in results from surgery. However, ECT eliminated the need for amputation and dysfunction often caused by a too wide surgical excision.
Wang reported on ECT for 74 cases of liver cancer with tumors ranging from 3 to 20 cm. in diameter. The treatments of 3 to 5 hours were repeated 2 to 5 times with 7 to 10 days between each treatment. Total remission rate was 63.51%. Best results were obtained with tumor diameters less than 9 cm. Additional use of cytotoxic drugs and embolization resulted in a 87.5% cure rate.
Song et al. treated 46 patients having thyroid adenoma with ECT and reported a 97.8% cure rate with a single treatment. This represents successful treatment of benign tumors and destruction of precancerous and early malignant changes.
The above reports from China vary in the amount of technical detail presented regarding each study. In general, however, the electrodes were inserted under local anesthetic. The number of electrodes depended upon the tumor size and shape. The goal was to encompass the tumor with the electric field. Xin et al. state that, depending upon tumor composition and location, soft, flexible or hard electrodes with 0.1 cm diameters were used. The anode(s) was(were) placed within the tumor and the cathode(s) was(were) separated by from 1-3 cm. from the anode(s) or by a distance of 2-3 tumor diameters. There were a minimum of 2 electrodes and, at the other extreme, 2 anodes and 4-6 cathodes set up in two groups to establish two electric fields for a tumor of 6 cm. or larger. The treatment time varied from 1.5-5 hours and the number of sessions ranged from 1 to 5, again depending upon tumor size and response to therapy. The voltage used averaged about 8V but ranged from 6 to 15 V. The current ranged from 40-100 mA and the number of coulombs delivered per session ranged from 250 to 2000° C. Quan gives a rule of thumb at 100° C. per 1 cm of tumor diameter. Song observed that, at 100° C., the area of destruction around the anode is 0.5-0.6 cm and the area around the cathode is 0.4-0.5 cm. Xin et al. observed some blockage of the heart beat in central lung cancer ECT with currents over 30 mA. Keeping the electrodes more than 3 cm from the heart corrected this effect.
The table below summarizes the types of tumors mentioned as having been treated by the researchers cited above: Author Tumor or Cancer Type Xin et al. Lung, squamous cell, esophageal, parotid, breast, sarcoma of the leg, skin, malignant melanoma, cartilage sarcoma of nose, thyroid, liver, keloid, angioma, freckle Matsushima Skin, breast, lung, gland et al. Song et al. Skin, malignant melanoma, lip, tongue, upper jaw parotid, breast, vagina, penis, osteogenic sarcoma, fibrosarcoma metastatic lymph node Lao et al. Liver (hepatocellular carcinoma, cholangiocellular carcinoma, mixed hepatocholangiocellular cancer, transparent liver cancer) Quan Soft tissue sarcoma, head/neck cancer, malignant melanoma, skin cancer, breast cancer, recurrent cancer, metastatic cancer Wang Liver
Animal Results
Yokoyama et al. used direct current in canine malignant cancer tissue and found that cancer tissues of 2 cm. in diameter around the electrode became necrotic in 60 minutes. Bleomycin was then injected intravenously and was found to accumulate around the electrode in the majority of cases. Li et al. studied the mechanisms of ECT in normal dog liver and verified that the cell metabolism and its environment are destroyed in agreement with previous theory. Chen et al. studied ECT in mice and verified much of the theory, including the conclusions that tumor cells are more sensitive to changes of their microenvironment than are normal cells and that ECT stimulates the immune system, pointing out that, at an electrode voltage as low as 100 mV, leukocytes concentrate at the anode and lymphocyte anti-tumor response might be activated. Li et al., like Xin, placed both an anode and a cathode in the tumor. Chou et al. investigated ECT in mice and rats. Pointing out that constant voltage is used in clinics to prevent pain, they used a constant-voltage mode. They also cite the observations of Xin that untreated tumors sometimes disappear after ECT of the primary tumor. The hypothesis proposed to explain this was that the immune system was enhanced by ECT. REFERENCES: - http://www.genetronics, retrieved Jul. 29, 2003.
- Electro-Cancer Treatment, http://www.st-georg.com/ect.html, retrieved Oct. 25, 1999.
- M. Belehradek, C. Domenge, B. Luboinski, S. Orlowski, J. Belehradek, Jr., L.M. MIR. Abstract of Electrochemotherapy, A new antitumor treatment. First clinical phase I-II trial. Cancer Dec. 15, 1993; 72(12):3694-700.
- K. Brandisky, I. Daskalov. Abstract of Electrical Field and Current Distributions in Electrochemotherapy, Biolectrochemistry and Bioenergetics Feb. 1999; 48(1):201-8.
- M. Cemazar, G. Sersa and D. Miklavcic. Electrochemotherapy with Cisplatin in the Treatment of Tumor Cells Resistant to Cisplatin, Anticancer Research 18: 4463-4466 (1998).
- S.L. David, D.R. Absolom, C.R. Smith, J. Gams, and M.A. Herbert. Effect of Low Level Direct Current on In Vivo Tumor Growth in Hamsters, Cancer Research 45, 5625-5631, Nov. 1985.
- D. Liu, Y.L. Xin, B. Ge, F. Zhao, H.C. Zhso. Experimental Studies on Electrolytic Dosage of ECT for Dog's Oesophageal Injury and Clinical Effects of ECT for Oesopohageal Anastomotic Opening Stenosis and Oesophageal Carcinoma, European Journal of Surgery 1994; Suppl 574: 71-72.
- R.A. Gatenby. Abstract of Mathematical Models of Tumour Invasion Mediated by Transformation-Induced Alteration of Microenvironment pH, Symposium 240: The Tumour Microenvironment: Causes and Consequences of Hypoxia and Acidity, p. 2-3, held at the Novartis Foundation, London, 240 Oct 10-12, 2000.
- L.F. Glass, N.A. Fenske, M. Jaroszeski, R. Perrott, D.T. Harvey, D.S. Reintgen, R. Heller. Abstract of Bleomycin-Mediated Electrochemotherapy of Basal Cell Carcinoma, Journal of the American Academy of Dermatology Jan. 1996; 34(1):82-6.
- H. Gong, G. Liu. Effect of Electrochemical Therapy on Immune Functions of Normal and Tumour-Bearing Mice, European Journal of Surgery, Suppl 1994; (574): 73-74.
- S.A. Grossman, P.S. Staats, Abstract of Current Management of Pain in Patients with Cancer. Oncology (Huntingt) Mar. 1994; 8(3):93-107.
- M.B. Habal. Abstract of Effect of Applied DC Currents on Experimental Tumor Growth in Rats, Journal of Biomedical Materials Research, vol. 14, 789-801 (1980).
- M.A. Hamza, P.F. White, H.E. Ahmed, E.A. Ghoname. Abstract of Effect of the Frequency of Transcutaneous Electrical Nerve Stimulation on the Postoperative Opioid Analgesic Requirement and Recovery Profile, Anesthesiology Nov. 1999;91(5):1232-8.
- C. Hauton, M. Charbonnier, L. Cara and J.P. Salles, A New Type of Liposome for Electrochemical Treatment of Cancer: The Lipogelosomes, European Journal of Surgery 1994; Suppl 574: 117-119.
- C.E. Humphrey, E.H. Seal. Biophysical Approach toward Tumor Regression in Mice, Science, vol. 130, 1959.
- M. Kraus and B. Wolf. Implications of Acidic Tumor Microenvironment for Neoplastic Growth and Cancer Treatment: A Computer Analysis, Tumor Biology 1996; 17: 133-154.
- M. Kraus and B. Wolf. Physicochemical Microenvironment as Key Regulator for Tumor Microevolution, Invasion, and Immune Response: Targets for Endocytotechnological Approaches in Cancer Treatment, Endocytobiosis & Cell Research, 12, 133-156 (1998).
- D. Miklavcic, D. An, J. Belehradek, Jr., L.M. Mir. Abstract of Host's Immune Response in Electrotherapy of Murine Tumors by Direct Current, European Cytokine Network Sep. 1997;8(3):275-9.
- D.M. Morris, M.D., A.A. Marino, Ph. D., and E. Gonzalez, M.D. Electrochemical Modification of Tumor Growth in Mice, Journal of Surgical Research 53, 306-309 (1992).
- E. Nilsson. Modelling of the Electrochemical Treatment of Tumours. Dissertation, Department of Chemical Engineering and Technology, Applied Electrochemistry, Royal Institute of Technology, Stockholm 2000.
- T. Nishi, S.B. Dev., K. Yoshizato, J. Kuratsu, Y. Ushio. Abstract of Treatment of Cancer Using Pulsed Electric Field in Combination With Chemotherapeutic Agents or Genes, Human Cell Mar. 1997;10(1):81-6.
- G.D. O'Clock, Ph. D. (E.E.), P.E. The Effects of In Vitro Electrical Stimulation on Eukaryotic Cells: Suppression of Malignant Cell Proliferation, Journal of Orthomolecular Medicine, vol. 12, No. 3, 1997.
- W.R. Panje, M.P. Hier, G.R. Garman, E. Harrell, A. Goldman, I. Bloch. Abstract of Electroporation Therapy of Head and Neck Cancer, Annals of Otology, Rhinology and Laryngology Sep. 1998; 107(9 Pt 1): 779-85.
- A. Plesnicar, G. Sersa, L. Vodovnik, J. Jancar, L. Zaletel-Kragelj and S. Plesnicar. Electric Treatment of Human Melanoma Skin Lesions with Low Level Direct Electric Current: An Assessment of Clinical Experience Following a Preliminary Study in Five Patients, European Journal of Surgery 1994; Suppl 574:45-49.
- N. Raghunand. Abstract of pH and Chemotherapy, Symposium 240: The Tumour Microenvironment: Causes and Consequences of Hypoxia and Acidity, p. 5-6, held at the Novartis Foundation, London, 240 Oct. 10-12, 2000.
- L.H. Ramirez, S. Orlowski, D. An, G. Bindoula, R. Dzodic, P. Ardouin, C. Bognel, J. Belehradek Jr., J-N Munck, and L.M. Mir. Electrochemotherapy on Liver Tumours in Rabbits, British Journal of Cancer (1998) 77(12). 2104-2111.
- M.K. Schauble, M.B. Habal. Electropotentials of Tumor Tissues. Journal of Surgical Research 9: 9, 1969.
- S. Seguchi, S. Kawauchi, Y. Morimoto, T. Arai, H. Asanuma, M. Hayakawa, M. Kikuchi. Abstract of Critical Parameters in the Cytotoxicity of Photodynamic Therapy Using a Pulsed Laser. Lasers Med Sci 2002, 17(4):265-71.
- G. Sersa, M. Cemazar, D. Miklavcic and D. J. Chaplin, Tumor Blood Flow Modifying Effect of Electrochemotherapy with Bleomycin, Anticancer Research 19: 4017-4022 (1999).
- B.N. Singh and C. Dwivedi. Antitumor Drug Delivery by Tissue Electroporation, Anti-Cancer Drugs 1999, 10, pp. 139-146.
- T.V. Taylor, P. Engler, B.R. Pullan and S. Holt. Ablation of Neoplasia by Direct Current, British Journal of Cancer (1994), 70, 342-345.
- A.L. Vandenbogaerde, E.M. Delaey, A.M. Vantieghem, B.E. Himpens, W.J. Merlevede, P.A. de Witte, Abstract of Cytotoxicity and Antiproliferative Effect of Hypericin and Derivatives After Photosensitization. Photochem Photobiol Jan. 1998;67(1):119-25.
- P. Vaupel, D.K. Kelleher, M. Hockel. Abstract of Oxygen Status of Malignant tumors: Pathogenesis of Hypoxia and Significance for Tumor Therapy. Semin Oncol Apr. 2001; 28(2 Suppl 8):29-35.
- L. Vodovnik, D. Miklavcic, G. Sersa. Modified Cell Proliferation Due to Electrical Currents, Medical and Biological Engineering and Computing, 1992, 30, CE21-CE28.
- H. von Euler, Electrochemical Treatment of Tumours, Doctoral Thesis, Swedish University of Agricultural Sciences, Uppsala 2002.
- J.C. Weaver. Electroporation: A General Phenomenom for Manipulating Cells and Tissues. J Cell Biochem 1993; 51 No. 4: 426-435.
- M. Wojcicki, R. Kostyrka, B. Kaczmarek, J. Kordowski, M. Romanowski, M. Kaminski, J. Klonek, S. Zielinski. Abstract of Electrochemical Therapy in Palliative Treatment of Malignant Dysphagia: A Pilot Study, Hepatogastroenterology Jan.-Feb. 1999;46(25):278-84.
- B. Wolf, M. Brischwein, W. Baumann, R. Ehret, T. Henning, M. Lehmann, A. Schwinde. Microsensor-Aided Measurements of Cellular Signalling and Metabolism on Tumor Cells, Tumor Biology 1998; 19:374-383.
- Y.L. Xin, F.Z. Xue, F.G. Zhao. Effectiveness of Electrochemical Therapy in the Treatment of Lung Cancers of Middle and Late Stage, Chinese Medical Journal 1997 110(5): 379-383.
- Y. Yen, J.R. Li, B.S. Zhou, F. Rojas, J. Yu and C.K. Chou. Electrochemical Treatment of Human KB Cells In Vitro, Bioelectromagnetics 20:34-41 (1999).
- Y.L. Xin, D. Liu. Electrostatic Therapy (EST) of Lung Cancer and Pulmonary Metastasis: Report of 15 Cases. European Journal of Surgery 1994; Suppl 574: 91-92.
- X.Z. Lin, C.M. Jen, C.K. Choud, D.S. Chou, M.J. Sung, T.C. Chou. Saturated Saline Enhances the Effect of Electrochemical Therapy. Digestive Diseases and Sciences 2000: 45(3): 509-514.
- Reis A. Henninger T. Zerstorung maligner Wachstumsenergie durch anodische Oxydation. Klin Wochenschrift 1951; 39.
- Nordenstrom B. Preliminary clinical trials of electrop horetic ionization in the treatment of malignant tumors. IRCS Med Sc 1978; 6: 537.
- Schanble MK, Mutaz HB, Gallick HD. Inhibition of experimental tumor growth in hamsters by small direct currents. Arch Pathol Lab Med 1977; 101: 294.
- Srinivasan S, Gahan Jr GL, Stoner GE. Electrochemistry in the biomedical sciences. In: Bloom H, Gutmann F (eds): Electrochemistry the last thirty and the next thirty years. New York: Plenum Press, 1977.
- Nordenstrom BEW. Biologically closed electri c circuits: clinical, experimental and theoretical evidence for an additional circulatory system. Stockholm: Nordic Medical Publications, 1983.
- Nordenstrom B. Biologically closed electric circuits: activation of vascular interstitial closed elec tric circuits for treatment of inoperable cancer. Journal of Bioelectricity 1984; 3(162): 137-153.
- Lao, Y., Ge, T., Zheng, X., Zhang, J., Hua, Y., Mao, S., Feng, X. Electrochemical therapy for intermediate and advanced liver cancer: a report of 50 cases. Eur J Surg 1994; Suppl 574: 51-53.
- Mir M, Orlowski S, Belehradek Jr J, Paoletti C. Electroctherapy potentiation of antitumor effect of bleomycin by local electric pulses. Eur J Cancer 1991; 27:68-72.
- Wolf B, Kraus M, and Sieben U, “Potential of microsensor-based feedback bioactuators for biophysical cancer treatment,” Biosensors and Bioelectronics, vol. 12, No. 4, pp 301-309, 1997.
- Kirsch DL, Lerner FN. Electromedicine: the other side of physiology. In: Innovations in pain management: a practical guide for clinicians. Winter Park, FL: GR Press, 1995.
- Li K, Xin Y, Gu Y, Xu B, Fan D, Ni B. Effects of direct current on dog liver: possible mechanisms for tumor electrochemical treatment. Bioelectromagnetics 1997; 18: 2-7.
- Berendson J, Simonsson D. Electrochemical aspects of treatment of tissue with direct current. Eur J Surg 1994: Suppl 547: 111-115.
- Song Y, Li C, Li Y, Song Q, Chang B, Song L, Liu C, Wang T. Electrochemical therapy in the treatment of malignant tumours on the body surface. Eur J Surg 1994; Suppl 574: 41-43.
- Matsushima Y, Takahashi E, Hagiwara K, Konaka C, Miura H, Kato H, Koshiishi Y. Clinical and experimental studies of anti-tumoural effects of electrochemical therapy (ECT) alone or in combination with chemotherapy. Eur J Surg 1994; Suppl 574: 59-67.
- Xin Y, Xue F, Ge B, Zhao F, Shi B, Zhang W. Electrochemical treatment of lung cancer. Bioelectromagnetics 1997; 18: 8-13.
- Nordenstrom BEW. Electrochemical treatment of cancer. I: variable response to anodic and cathodic fields. Am J Clin Oncol (CCT) 1989; 12(6): 530-536.
- Nordenstrom BEW. Survey of mechanisms in electrochemical treatment (ECT) of cancer. Eur J Surg 1994; Suppl 574: 93-109.
- Chen B, Xie Z, Zhu F. Experimental study on electrochemical treatment of cancer in mice. Eur J Surg 1994; Suppl 574: 75-77.
- Chou C, McDougall JA, Ahn C, Vora N. Electrochemical treatment of mouse and rat fibrosarcomas with direct current. Bioelectromagnetics 1997; 18: 14-24.
- Nordenstrom BEW, Eksborg, S., Beving, H. Electrochemical treatment of cancer. II. effect of electrophoretic influence on adriamycin. Am J Clin Oncol (CCT) 1990; 13(1): 75-88.
- Xin, Y. Organisation and spread of electrochemical therapy (ECT) in China. Eur J Surg 1994; Suppl 577: 25-30.
- Quan, K. Analysis of the clinical effectiveness of 144 cases of soft tissue and superficial malignant tumours related with electrochemical therapy. Eur J Surg 1994; Suppl 574: 37-40.
- Wang, H. Electrochemical therapy of 74 cases of liver cancer. Eur J Surg 1994; Suppl 574: 55-57.
- Song, L., Liu, C., Zhang, B., Wang, T., Song, Y., Li, Y. Electrochemical therapy (ECT) for thyroid adenoma during acupuncture anaesthesia: analysis of 46 patients. Eur J Surg 1994; Suppl 574: 79-81.
- Yokoyama, M., Itaoka, T., Nakajima, H., Ikeda, T., Ishikura, T., Nitta, S. [The use of direct current in the local destruction of cancer tissues]. Gan To Kagaku Ryoho Apr. 1989; 16(4 Pt 2-2): 1412-1417.
- Okino, M. and Mohri, H. Effects of high voltage electrical impulse and an anti-cancer drug on growing tumors. Japanese Journal of Cancer Research, vol. 78, pp. 1319-1321, 1987.
- Orlowski, S., Belehradek, J.J. Paoletti, C. and Mir, L.M. “Transient electropermeabilization of cells in culture increase of the cytotoxicity of anti-cancer drugs”, Biochem, vol. 37, No. 24, pp. 4727-4733, 1988.
- Belahradek, J.J., Orlowski, S., Raimiriz, L.H., Pron, G., Poddevin, B. and Mir, L.M., “Electropermeabilization of cells and tissues assessed by the qualitative and quantitative electroloading of bleomycin”, Biochem. Biophys. Acta, vol. 1190, pp. 155-163, 1994.
- Hofmann, G.A., Dev, S.B., Dimmer, S. and Nanda, G.S., “Electroporation Therapy: A new approach to the treatment of head and neck cancer, IEEE Transactions On Biomedical Engineering”, vol. 46, No. 6, pp. 752-759, 1999.
- Sersa, et al. Improvement of Combined modality therapy with cisplatin and radiation using electroporation of tumors. Int. J. Radiation Oncology Biol. Phys. vol. 46, No. 4:1037-1041. (2000).
- Hofmann, Dev, Nanda, and Rabussay. electroporation therapy of solid tumors. Critical Reviews in therapeutic Drug Carrier Systems 16(6):523-569 (1999).
- Samuelsson, Harnek, Ewers, Jonsson. Electrochemical and megavolt treatment of rat tumors. Eur J Surg Suppl 574:69-70. (1994).
- Habal and Schauble. An implantable CD power unit for control of experimental tumor growth in hamsters. Medical Instrumentation 7 No. 5: 305-306. (1973).
- Semrov and Miklacic. Calculation of the electrical parameters inn electrochemistry of solid tumors in mice. Comp Biol Med 28:439-448. (2000).
- Turler, Schaefer, et al. Local treatment of hepatic metastases with low level direct electrical current: experimental results. Scand J Gastroenterol. 3:322-328. (2000).
- Schecter, DC. “Containment of Tumors Through Electricity.” PACE 1979. vol. 2, pp. 100-114.
- Damascelli B, Patelli G, Frigerio LF, Lanocita R, Di Tolla GD, Marchiano A., Spreafico C, Garbagnati F, Bonalumi MG, Monfardini L Ticha V, Prino A. First clinical experience with a high-capacity implantable infusion pump for continuous intravenous chemotherapy. Cardiovasc Intervent Radiol 1999; 22: 37-43.
- Ranade VV. Drug delivery systems. 4. Implants in drug delivery. J Clin Pharmacol 1990; 30 No. 10: 871-889.
- Buchwald H, Rohde TD. Implantable pumps. Recent progress and anticipated future advances. ASAIO J 1992; 38 No. 4: 772-778.
- Wigness BD, Dorman FD, Robinson Jr HJ, Arendt EA, Oegema Jr TR,Rohde TD, Buchwald H. Catheter with an anchoring tip for chronic joint capsule perfusion. ASAIO Trans. 1991; 37 No. 3: M290-292.
- Heruth KT, Medtronic SynchroMed drug administration system. Ann NY Acad Sci 1988; 531: 72-75.
- Vogelzang NJ, Ruane M, DeMeester TR. Phase I trial of an implanted battery-powered, programmable drug delivery system for continuous doxorubicin administration. J Clin Oncol 1985; 3 No. 3: 407-414.



|


