

The million dollar question: Does adding hydrogen/oxygen to a car engines intake increase mileage?
Here's the five main half-truths that are being used by slick salesmen to sell hydrolysers supposedly to boost car mileage;
Half Truth: Stanley Meyers made a water powered car but was knocked off before he could make a conversion kit.
Whole Truth: Meyers had to return $25,000 to his investors after a court ruled that his claims, by which he obtained the investors, were fraudulent. He couldn't demonstrate his water-car claims to his investors and so they asked for their money back. He wasn't gentleman enough to do that and so they took him to court where he was given four opportunities to demonstrate his water car to qualified men of science but he always made excuses instead of showing it. Maybe someone that hates scam artists poisoned him but the coroners report said he died of a cerebral aneurysm (blood vessel bursting in the brain). The only truth to this half-truth is that he died before he could make a kit (by which he would of scammed thousands of people). [see Wikipedia page]
Half Truth: Hydrogen/oxygen (hydroxy) from an on-board water electrolysis unit (hydrolyser) is a fuel.
Whole Truth: Yes it is but if your car has a modern alternator then the engine energy required to make the electricity (via the alternator) is more than the energy derived from combustion of the hydrogen. Therefore in this case it is not useful as a fuel. It would have to be stored in a pressurized tank so that no car energy is being used to produce it. Mr. George Vosper, Professor of Engineering, said: “it will certainly take more power to produce this hydrogen than can be regained by burning it in the engine. i.e. the resulting energy balance should be negative. If the aim is to create hydrogen by electrolysis to be burned as a fuel, the concept is ridiculous.” Only the old style alternators do not create more engine load with more electrical load. (read more).
Half Truth: Adding hydroxy to the engine intake will cause the gasoline in the cylinders to combust more rapidly.
Whole Truth: 2 LPM hydroxy will cause the fuel combustion to happen less than 3 percent more rapidly. The study report "Laminar Flame Speeds of Hydrocarbon and Air Mixtures" in Combustion and Flame Journal (vol 63, 1986, pp 339-347) has test graphs (see below) that show added hydrogen at different percentages of the fuel energy (.1=10%, .2=20% etc) and the resultant increase in flame speed. You can see that adding 10% of stoichiometric gaseous hydrogen/air only increases the flame speed of the two hydrocarbon fuels by an average (between the two tests) of 17%. If the flame front speed of gasoline/air (being also a hydrocarbon fuel) was increased by 17% then that would increase the engine efficiency by around 21% (assuming that most of the work is done before 45° ATDC). But let's see what percentage of the intake a 2 LPM hydrolyser provides. First the 2 LPM hydroxy is equivalent to 4.4 LPM stoichiometric hydrogen/air since hydroxy's hydrogen concentration is 2.2 times more. (Stoichiometric hydrogen/air is 30% hydrogen, and hydroxy is 66% hydrogen. A stoichiometric mixture is 2 parts hydrogen to 1 part oxygen.) A typical economy cars intake is around 350 LPM of air. 4.4 divided by 350 is .0126 which is only 1.26% of the total engine intake. 1.26 divided by 9 gives .14 which is 14% of the studies tested 9% intake volume of added hydrogen/air. Then 14% of 20 is 2.8 which means that 2 LPM hydroxy will increase flame speed by 2.8% which is almost nothing at all. It would take 31 LPM of stoichiometric gaseous hydrogen/air to equal 9% volume of the engine intake to cause a 20% increase in flame front speed. This would equate to 14 LPM of hydrogen/oxygen from a hydrolyser to have the same amount of hydrogen. So you'd have to install seven 2 LPM hydrolysers to get 14 LPM hydroxy. But then that would cause the alternator to be a much greater load on the engine which would turn that 21% efficiency gain to probably less than 10%. And you'd probably have to install a heavier duty alternator to handle the excessive electrical current drain needed by seven hydrolysers.
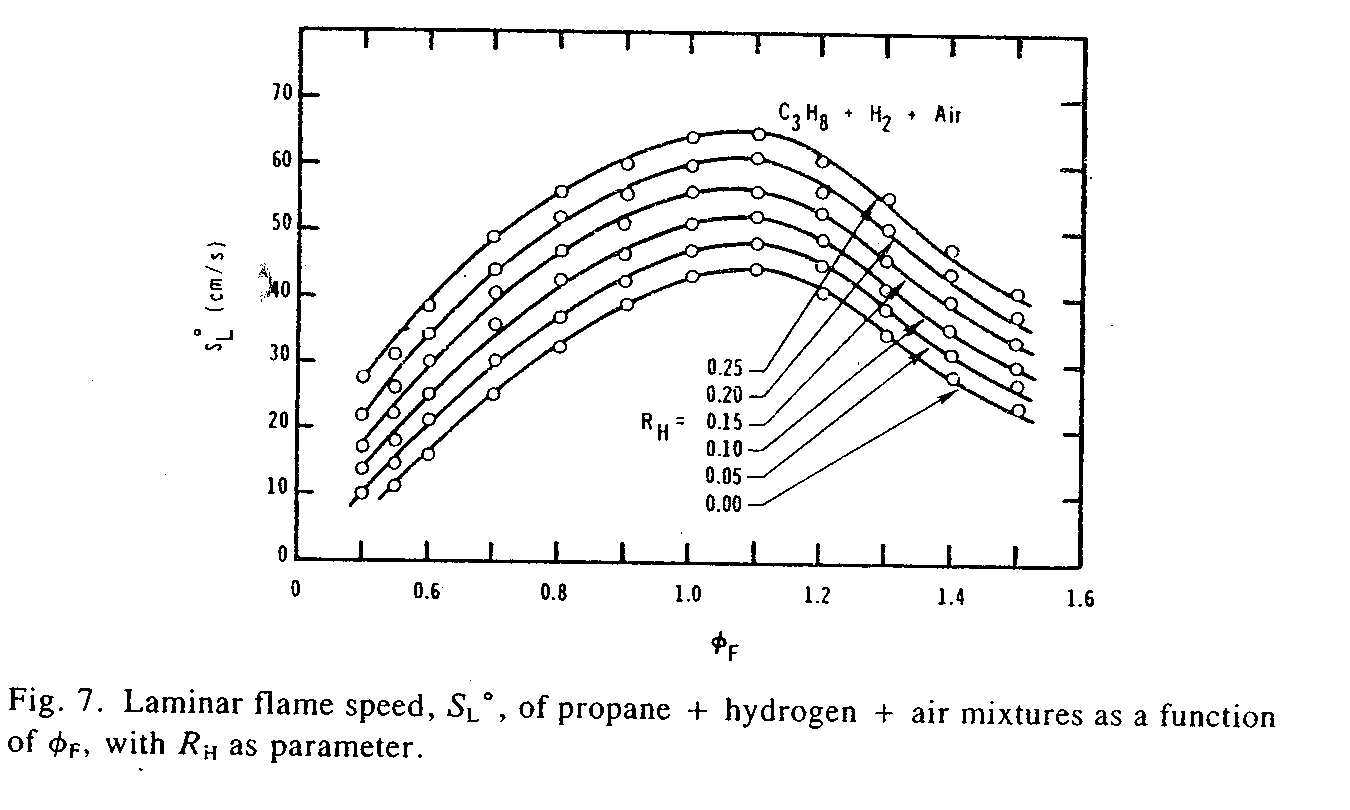
SL=laminar flame speed in cm/sec. øF=change of air/fuel (1.0 being normal stoichiometric, 1.4 being 40% leaner, 0.6 being 40% richer), C3H8=propane. CH4=methane. H2=hydrogen. RH=volumetric ratio of stoichiometric hydrogen/air divided by stoichiometric gasoline/air so that RH is the hydrogen/air fraction of the gasoline/air. (0.1 is 1 part hydrogen/air to 10 parts gasoline/air which is equal to 9.1% of the total)
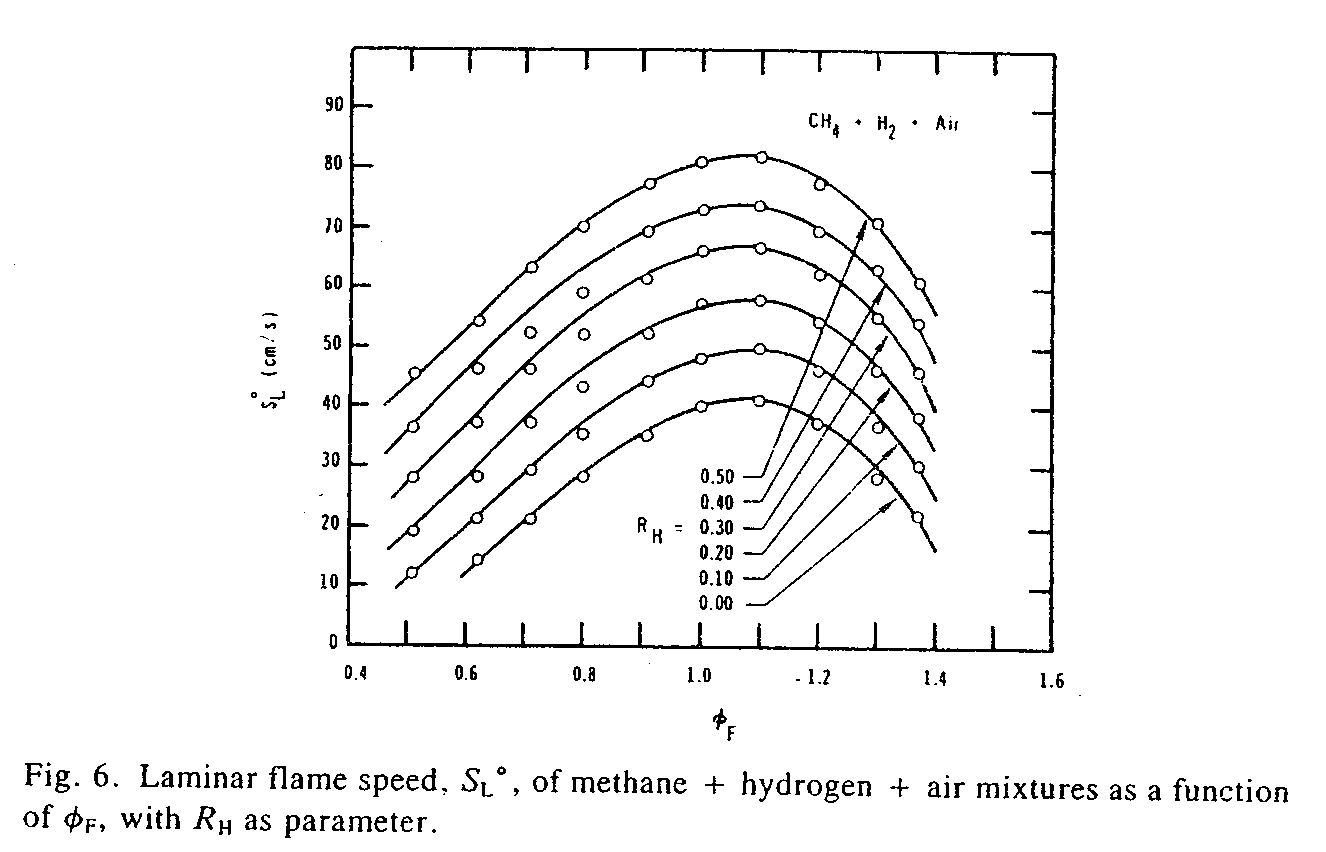
In the above graph for methane an RH of 0 yields 40.5cm/sec flame speed, and an RH of .1 yields a 47cm/sec flame speed which is a 16% increase. And for propane an RH of 0 yields 43cm/sec flame speed, and an RH of .1 yields a 51cm/sec flame speed which is a 18.6% increase. The average of the two increases is 17.3%.
Half Truth: Hydrogen/oxygen from a hydrolyser will automatically cause your car to get much better mileage.
Whole Truth: Scientists and researchers have been experimenting and writing reports on ways to use hydrogen in engines for decades now. But in reviewing the studies I only find one use for hydrogen when used as a supplement to gasoline and that is to assist the combustion of a leaner-than-normal air/fuel mixture. According to SAE paper #2007-24-0030 "Lean SI Engines: the Role of Combustion Variability in Defining Lean Limits" very lean mixtures (of low gasoline content) have a slower-than-normal burn rate (30% leaner takes 37% longer to burn 80% of the fuel ) and has varying flame speeds over +40% lean which gradually become more unstable until the engine starts to misfire at +78% lean. The presence of enough hydrogen (from liquid) insures that the gas will ignite every time and will "fill in the gaps" caused by a lean mixture to speed up the otherwise slow burn rate. The leanness of the mixture is what provides better gas mileage, but only if there's no constant misfiring. Liquid hydrogen is a great assist to a gas saving system. But hydrolysers provide little benefit for gas mileage because the actual amount of hydrogen they produce is just too small. When it comes to hydrogen addition the only proven gas saving system is adding hydrogen (from liquid) along with a means to achieve a leaner air/fuel ratio. The following is a key graph from their report which shows no significant increase of engine efficiency by hydrogen addition without additionally causing the fuel mixture to be leaner. The left side of the graph, at 1.0 Lambda, is the normal air/fuel ratio. With and without hydrogen approximately the same engine efficiency results. Efficiency increases by 13% at 60% leaner, with or without hydrogen.
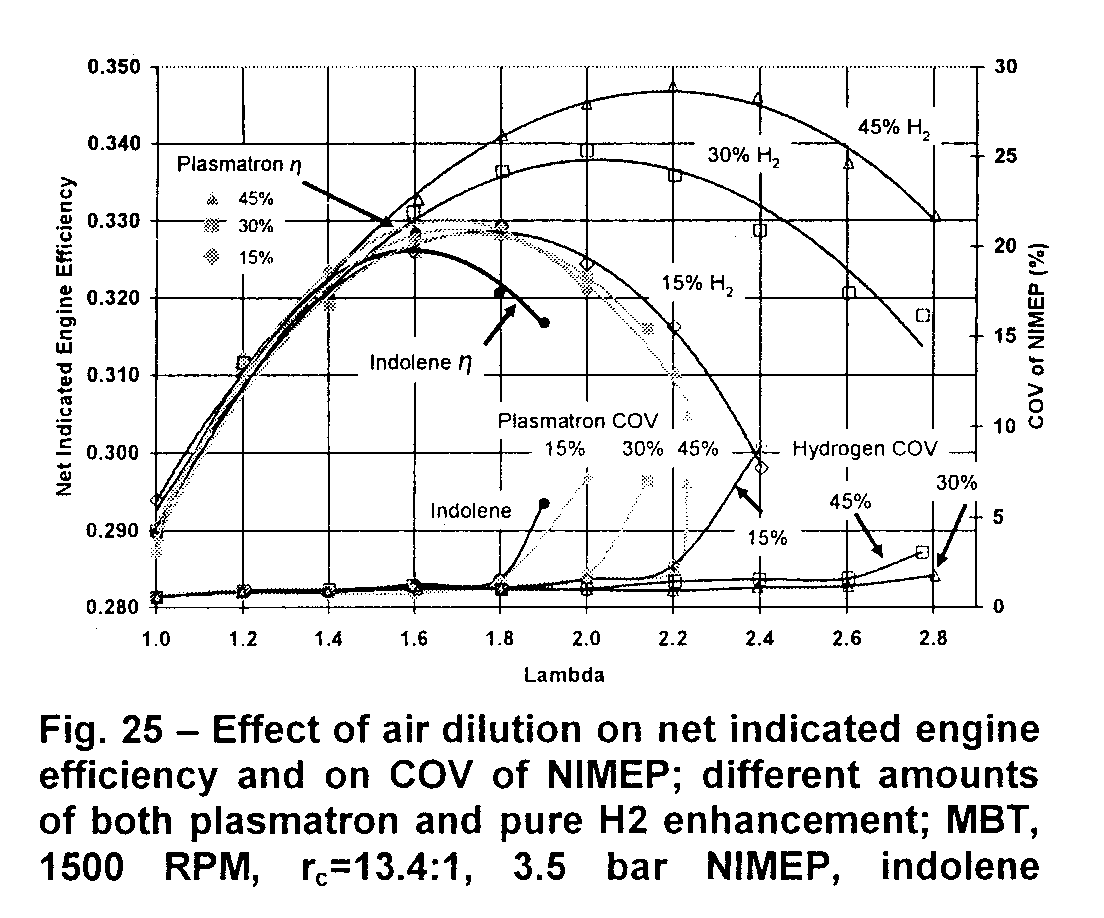
H2=pure hydrogen. Indolene is a standardized gasoline that researchers use. Its curve here is for air/indolene as fuel. COV is "coefficient of variability" of fuel mixture ignition (how much the moment of true fuel ignition varies from when the spark happens). Lambda is in reference to the air/fuel ratio so that 1.6 is 60% more air than the standard fuel/air mixture. 15% H2 is 15% of the total fuel energy derived from hydrogen which equates to around 60 LPM hydroxy for a 1.8L engine at 2000 rpm.
Read and study the reports to see for yourself that hydrogen is only an upper lean limit aid. Here are some good quotes from the research papers I've obtained:
Title: "Lean Air-Fuel Mixtures Supplemented With Hydrogen For S. I. Engines: A Possible Way To Reduce Specific Fuel
Consumption?" by R. Gentili
Source: Journal of Hydrogen Energy, vol. 10, pp 491-495, 1985
Quotes:
"With a homogenous charge*, the presence of hydrogen in the mixture increases combustion speed and reduces misfiring. As a
result, the lowest specific fuel consumptions are obtained with leaner mixtures as a whole. Experimental works show the lean
operating limit** increases in a nearly linear way with the increase of the hydrogen fraction. An energy ratio of .45*** may
represent the maximum hydrogen supplementation level of practical interest."
* (mixture of gasoline and hydrogen)
** (limit of leanness of gasoline that still insures normal operation)
*** (supplied hydrogen energy divided by gasoline energy=.45 which equates to 120 LPM hydroxy for my Honda Accord)
Title: "Performance and Fuel Consumption Estimation of a Hydrogen Enriched Gasoline Engine at Part-Load Operation" by G.
Fontana, E. Galloni, E. Jannelli and M. Minutillo
Source: Dept of Industrial Engineering, University of Cassino (January, 2002). SAE Technical Paper Series #2002-01-2196
Quotes:
"Under most loads near-stoichiometric air/fuel mixtures* are still required for normal acceleration, although under idle
conditions, reduced loads** and moderate acceleration hydrogen addition in combination with lean burn engine conditions can
guarantee a regular running of the engine with many advantages in terms of emissions levels and fuel consumption."
"The hydrogen supplementation increases the combustion speed and extends the equivalence ratio lean-limit of the mixture
reducing the misfiring. Thus, operation at ultra-lean air-gasoline mixtures is possible"
"The idea is that a small hydrogen amount can permit leaning the gasoline-air mixture."
* (near 14.6:1 ratio by weight)
** (reduced engine load: cruising)
The studies quoted above only added liquid hydrogen, not hydrogen/oxygen like what you get from a hydrolyser, to the air/fuel mixture. Skeptics to these conclusions have said that the studies weren't valid because of the lack of added oxygen. But consider this: There is plenty of extra oxygen for the hydrogen to combust with that will not be used in the combustion of the gasoline. This is evident by the fact that fuel injected cars can detect oxygen in the exhaust by the oxygen sensor. Here is a study that directly addresses that theme: "Investigating Combustion Enhancement and Emissions Reduction With the Addition of 2hD2 + OD2 to a SI Engine" SAE paper #2003-32-0011 "Further, the addition of hydrogen only and hydrogen and oxygen in a ratio of 2:1 were compared. The extra oxygen had little effect on engine performance other than an increase in NO exhaust concentration" (NO is Nitrous Oxides)
Half Truth: Hydrogen/oxygen from a hydrolyser will automatically cause your car to emit less of all the exhaust pollutants.
Whole Truth: Actually it will cause more pollution (nitrous oxides) unless you lean the fuel ratio. The research papers reveal that all of the main pollutants will be reduced only if the air/gasoline mixture is leaner than normal. Otherwise the extra heat at the top of the combustion stroke that is caused by the extra fuel of the hydrogen and the extra pressure from its combustion will create more nitrous oxides which are a combination of nitrogen (the most plentiful gas in air) and oxygen that combine when there's high heat, which is caused by adding hydrogen. A lean air/fuel mixture counters that by the reduction of fuel and the subsequent reduction of peak heat. From the research paper Performance and Fuel Consumption Estimation of a Hydrogen Enriched Gasoline Engine at Part-Load Operation; "Because of the relationship between NOx (nitrous oxide) formation and temperature inside the cylinder, an increase of NOx concentration characterizes mixtures with high amount of hydrogen (faster burning rate). However, in Figure 2, a lesser NOx concentration for hydrogen-gasoline-air mixtures than for gasoline-air mixtures can be noted, except in correspondence of high torque values (60 Nm and 75 Nm). The reason for a low concentration is in the air overabundance (driving a wide open throttle and at lean mixtures) that limits the peak temperatures. Obviously, it is possible to mark (Figure 3) a decrease of exhaust carbon dioxide, according to low gasoline equivalence ratios." (Carbon dioxide only lowers with leaner mixtures of air/gasoline. The "high torque values" mentioned are the high engine loads during acceleration.)
Both carburetor equipped cars and fuel injected cars average around a 14.6-to-1 air to gasoline ratio (by weight). This ratio is only necessary in reality when accelerating. When cruising or decelerating the air/fuel ratio can be much leaner for more gas mileage, as much as 40% leaner.
Only above 78% leaner will there be much misfiring (occasionally not igniting the air/fuel mixture) which leaves much unburned gasoline in the exhaust which is damaging to the catalytic converter [1] and is very polluting. Misfires happen because very lean air/fuel ratios can fail to ignite from the spark of the spark plug, and when there's ignition the combustion will have a much slower flame front speed [2] so that some gasoline vapors are expelled through the valved exhaust port before they have a chance to burn. This slow flame front also causes much less horsepower because ideally all the gasoline will burn before the piston has gone 45 degrees through its power stroke, during which is where it has the maximum combustion pressure (peaking around 20 degrees after top dead center, close to the top) to push against the piston and crank for engine power. [3]
To cure the misfire/pollution/low-power problem caused by a very lean fuel mixture (+78% lean) you would have to introduce liquid hydrogen. It will increase the speed at which the lean air-gasoline mixture burns in order to restore lost horsepower, prevent misfiring, and reduce exhaust pollutants (carbon monoxide, carbon dioxide, nitrogen oxides). The study report "Laminar Flame Speeds of Hydrocarbon and Air Mixtures" in the Combustion and Flame Journal said that a 30% lean mixture takes twice as long to burn 100% of the fuel and that returning it to normal would require around 18% volume stoichiometric hydrogen/air.
Unfortunately many uneducated and/or unscrupulous internet vendors are promoting the idea that everyone can get better gas mileage just by adding hydroxy to your cars intake. But there is no scientific literature to support these claims, just a bunch of made-up "testimonies". There are plenty of scientific reports and studies proving that a lean air/fuel mixture can increase mileage and reduce exhaust pollutants, and that added liquid hydrogen can allow even more engine efficiency. Here are a few:
Houseman J, "Lean Combustion of Hydrogen Gasoline Mixtures". Abstracts of papers of the American Chemical Society (169): 6-6 1975.
Hoehn F.W., Baisley R.L., Dowdy M.W., "Advances In Ultralean Combustion Technology Using Hydrogren-Enriched Gasoline", IEEE Transactions on Aerospace and Electronic Systems 11 (5): 958-958 1975
Gentil R., "Lean Air-Fuel Mixtures Supplemented with Hydrogen for S.I. Engines: A Possible Way To Reduce Specific Fuel Consumption?", Int'l Journal of Hydrogen Energy, vol. 10, No.7/8, pp. 491-495, 1985
May H., Gwinner D., "Possibilities of Improving Exhaust Emissions and Energy Consumption in Mixed Hydrogen-Gasoline Operation", Int'l Journal of Hydrogen Energy, vol. 8, No. 2, pp. 121-129, 1982
In the early eighties George Vosper P. Eng., ex-professor of Dynamics and Canadian inventor, designed and patented a device to transform internal combustion engines to run on hydrogen. He later affirms: “A small amount of [liquid] hydrogen added to the air intake of a gasoline engine would enhance the flame velocity and thus permit the engine to operate with a leaner air to gasoline mixture than otherwise possible. The result, far less pollution with more power and better mileage.”
References:
[1] http://courses.washington.edu/me341/oct22v2.htm
[2] Performance and Fuel Consumption Estimation of a Hydrogen Enriched Gasoline Engine at Part-Load Operation, SAE paper # 2002-01-2196 "In fact, the addition of small hydrogen quantities increases the flame speed at all gasoline equivalence ratios, so the engine operation at very lean air-gasoline mixtures is possible."
[3] http://en.wikipedia.org/wiki/Internal_combustion_engine "The thermodynamics of the idealized Carnot heat engine tells us that an ICE is most efficient if most of the burning takes place at a high temperature, resulting from compression—near top dead center. The speed of the flame front is directly affected by the compression ratio, fuel mixture temperature, and octane or cetane rating of the fuel. Leaner mixtures and lower mixture pressures burn more slowly requiring more advanced ignition timing."
See this video showing the combustion lasting till the exhaust valve opens. This is accurate according to this quote from a physicist: "In an engine that is operating properly, contributions to this Integral begin at the instant of ignition and end when the exhaust valve begins to open" from http://mb-soft.com/public2/engine.html
Modifying Your Car to Consume Less Gasoline
For fuel injected cars the hydrolyser vendors want to sell you an EFIE (electronic fuel injection enhancer), a cheap electronics circuit that splices into the oxygen sensor wiring, for $60. What they won't tell you is that at maximum it can only lean the air/fuel ratio 2.5% which isn't much. Standard oxygen sensors have a narrow band of response in which their voltage changes from .2 volts at the air/fuel ratio of 14.9:1 or leaner, to .8 volts at the air/fuel ratio of 14.2:1 or richer. The maximum variation from the middle ratio of 14.55:1 is 2.5% which equates to only around 2.5% gas savings. There is a way to lean down the fuel mixture in both carbureted and fuel injected cars though, up to 30% or more.
Click here for the next page